The most important point of the future chemical management system in Vietnam is the development of an inventory for existing chemical substances and the regulations for new chemical substances, but as of April 2021, the specific regulations have not been announced by government officials. Therefore, the following is an explanation of what has been done and what is planned for the future.
The Law on Chemicals (06/2007/QH12) already mentioned the National Chemical Inventory (NCI) and the registration requirements for new chemical substances related to it. Later in 2012, the “Prime Minister’s Decision approving the proposal for the creation of a NCI and National Chemical Database (768/QD-TTg)” was promulgated. In this decision, the period of 2012-2015 is divided into two phases, and the actions in each phase are specified as follows: but, in fact, only the National Chemical Database was completed now:
| Phase | Details |
|---|---|
| 1st Phase (2012-2014) |
|
| 2nd Phase (2015) |
|
*However, in fact, only the National Chemical Database was completed now. The NCI is yet under development by Vinachemia, MOIT.
On September 15, 2016, the draft of the NCI was released by the MOIT for public comment. The first draft listed 3023 substances, and with the aim of expanding the information on the list, the Vietnam Chemical Agency (Vinachemia) conducted a first call for opinions for chemicals not listed in the draft until October 30, 2016.
Additional calls have been conducted in November 2017, September 2018, April 2020, and October 2020. In particular, the latest nomination, which started in October 2020, attracted a lot of attention as it lasted for a long period of six months. However, it is important to note that the numbers listed below are simply the number of substances submitted, not the number of substances approved in NCI. In order for a substance to be approved in the inventory, it must pass a review by the Vinachemia. The process of the review is not disclosed, but it is assumed that the validity or completeness of the SDS and various evidence documents submitted are checked.
-
- 1st Draft: Sep, 2016 — 4,927 substances
- 2nd Draft: Nov, 2017 — 16,000 substances
- 3rd Draft: Sep, 2018 — 31,745 substances
- 4th Draft: Apr, 2020 — 36,777 substances
- 5th Draft: Oct, 2020 — 41,307 substances

The following figure shows the relationship between the list of current regulated chemical substances in Vietnam and the NCI. Vietnam’s Law on Chemicals stipulates management requirements based on the hazards of chemical substances, and the newly prepared inventory will be a list of chemical substances that are already in circulation in Vietnam. And, substances that are not listed in this inventory, that is NCI, will be treated as new chemical substances.
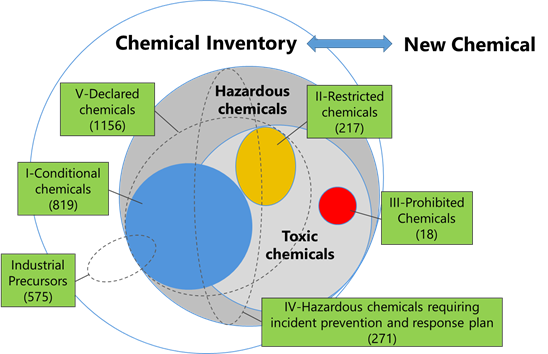
I. Conditional Chemical (819), Industrial Precursors (575)
II. Restricted Chemicals (217)
III. Prohibited Chemicals (18)
IV. Hazardous Chemicals requiring incident prevention and response plan (271)
V. Declared Chemicals (1156)
In addition, new chemical substances under the Law on Chemicals are classified into two categories, as shown below. The former one is completely new substances and the later one is called foreign chemical substances. Before distribution and use of new chemical substances in Vietnam, it is essential to obtain “New Chemical Substance Registration Approval Form” issued by the competent authority through a management procedure.
“New chemical”
A chemical not yet listed in the national chemical inventory (NCI) or foreign chemical inventories recognized by Vietnamese competent state agencies
“Foreign chemical”
A chemical not yet listed in the nation chemical inventory (NCI) but already listed in foreign chemical inventories recognized by Vietnamese competent state agencies
EnviX Ltd., a developer of Enviliance ASIA, can support NCI procedure and provide consulting service relating to chemical regulations of Vietnam. Please feel free to contact us:
Contact Form
 Chemical inventory development in Vietnam
Chemical inventory development in Vietnam

























